|
Listen to this article
|
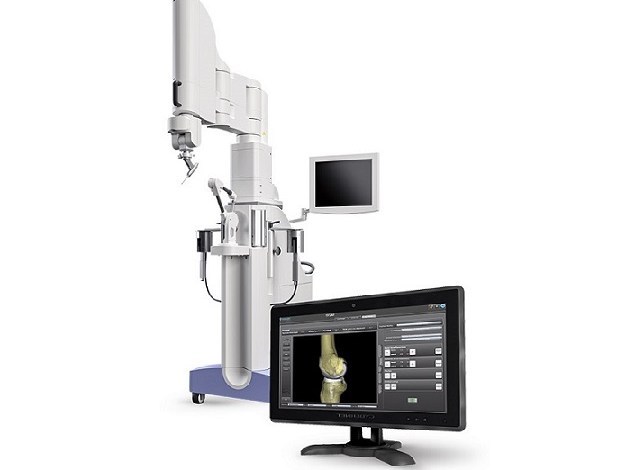
Think Surgical’s TSolution One robotic-assisted surgical system for total knee application.
The U.S. Food and Drug Administration (FDA) granted 510(k) clearance for the second generation of Think Surgical‘s TSolution One total knee application. The TSolution One is an active robot for total knee replacement. It includes fully automated bone preparation and provides surgeons a choice of implant options.
The newly-cleared version of the system features includes an enhanced pre-surgical planning user interface, quick-change tooling, improved surgeon accessories, and advanced bone model generation, according to Think Surgical.
FDA cleared the first generation of the robot more than a year ago. Based in Fremont, Calif., Think Surgical raised a funding round worth $134 million in March 2019.
“The ongoing evolution of the TSolution One total knee application is a testament to Think Surgical’s dedication and investment in advancing the use of robot technology in the orthopedic setting,” said Think Surgical’s acting CEO Jay Yang said in a news release. “The versatile, open platform provides surgeons with the flexibility of using a variety of implants, while offering hospitals and ambulatory surgery centers a sustainable, high throughput system for their ever-increasing total knee replacement procedures.”
In other healthcare robotics news, Johnson & Johnson (J&J) yesterday shared details about its new Ottava surgical robotic platform. The Ottava system has six arms to provide more control and flexibility in surgery, while its arms will be integrated into the operating table. J&J claimed Ottava has a zero-footprint design to enable patient access, increase space in the operating room, and improve workflow. J&J acquired Auris Health in 2019 for $3.4 billion and brought on Dr. Fred Moll as its chief development officer.
Robot-assisted surgery is currently a hot area in medical technology, with J&J, Medtronic and others seeking to take on Intuitive Surgical, whose da Vinci robot is the market leader. J&J claimed that Ottava will offer more flexibility than any currently available system.






Tell Us What You Think!