|
Listen to this article
|
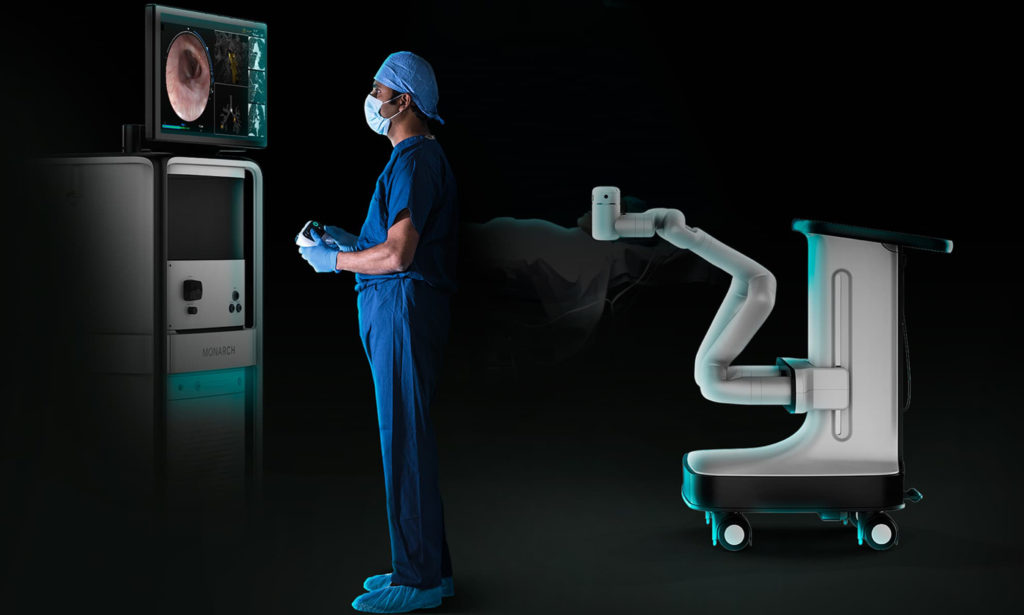
Johnson & Johnson now owns Auris, which makes the Monarch endoscopy platform. Source: Auris Health
Johnson & Johnson Services Inc. said in its quarterly earnings call yesterday that it has decided not to file immediately for regulatory approval for its planned general surgical robot. The New Brunswick, N.J.-based company said it now plans to start the first in-human studies for the system by the second half of 2022.
The decision not to follow a U.S. Food and Drug Administration (FDA) 510(k) clearance pathway for its robot was one of the major pieces of information to come out of Johnson & Johnson’s earnings call.
Before the COVID-19 pandemic affected the U.S., the company had been planning to reveal its surgical robot plans in May. The medical technology giant has been working on combining technologies that came out of its previous Verb Surgical collaboration with Verily, a life-sciences unit of Google parent Alphabet Inc. — as well as its $3.4 billion purchase of Auris Health Inc. and its FDA-cleared Monarch platform.
Johnson & Johnson, others pursue robot surgery market
Robot-assisted surgery is currently a hot area in healthcare, with with Johnson & Johnson, Medtronic PLC, and others seeking to take on Intuitive Surgical Inc., the dominant company in the space.
“We continue to be impressed by the technology advancements we’re seeing with both the Verb and Verily and the Auris combination,” said Alex Gorsky, CEO of J&J, during the quarterly earnings call. Our teams are making very good progress as we speak. … And we’ll be providing you some further updates regarding a more focused digital review in the coming months.”
Development of surgical robotics systems is challenging from both technology and regulatory perspectives. Titan Medical Inc. this week announced that it is resuming development of its Sport system after delays while it searched for funding.
Editor’s note: For more news on medical devices, visit MassDevice.com, a sibling site to The Robot Report.





Tell Us What You Think!