Sitting in New York City, looking up at the clear June skies, I wonder if I am staring at an endangered phenomena. According to many in the unmanned aircraft systems (UAS) industry, skylines across the country soon will be filled with flying cars, quadcopter deliveries, emergency drones, and other robo-flyers. Moving one step closer to…
Corindus CorPath GRX surgical robot receives clearance in Japan
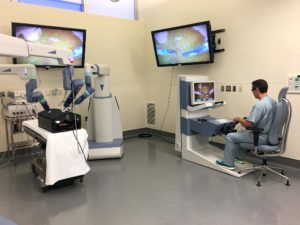
Corindus Vascular Robotics has won clearance from Japan’s Pharmaceutical and Medical Device Agency for its CorPath GRX robotic surgical device. The CorPath GRX vascular robotics platform is designed to assist cardiologists in performing percutaneous coronary interventions (PCI) procedures, including angioplasty and stent placement. Japan is one of the largest markets in the world for PCIs, according to…
TransEnterix Senhance surgical robot gets approved for new procedures
TransEnterix has won expanded indications from the Food and Drug Administration for its Senhance robot-assisted surgical system. The system is now cleared for hernia and gallbladder procedures. There are approximately 760,000 inguinal hernia and 1.2 million laparoscopic cholecystectomy procedures performed annually in the U.S. With this clearance, the system’s total addressable annual procedures in the…
Liability in robotics: inside the legal debate
I was dumbfounded by the XPONENTIAL 2018 keynote speech by Professor Zeynep Tufekci of the University of North Carolina. To paraphrase, ‘In the future, we will no longer need two pilots, planes will have just one captain and a dog. The dog will be there to bite the human in case he touches anything.’ Walking the…
Regulatory Challenges Holding Back Healthcare AI
It was the last question of the night, and it hushed the entire room. An entrepreneur expressed his aggravation about the US Food and Drug Administration’s (FDA) antiquated regulatory environment for AI-enabled devices to Dr. Joel Stein of Columbia University. Stein, a leader in rehabilitative robotic medicine, sympathized with the startup, knowing that tomorrow’s exoskeletons…
FDA Clears Software for Corindus CorPath GRX Surgical Robot
Corindus Vascular Robotics has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the first automated robotic movement designed for its CorPath GRX platform. Called “Rotate on Retract” (RoR), the proprietary software feature is the first automated robotic movement in the technIQ Series for the CorPath GRX platform. It allows the operator…
Honda wins CE Mark for Walking Assist Device
Honda (NYSE:HMC) said yesterday it won CE Mark approval in the European Union for its Honda Walking Assist Device designed for walking rehabilitation. The walking assist device uses cooperative control technology, based on Honda’s cumulative study of human walking, the Japanese company said. Read the whole story on our sister site, MassDevice.com
Procept BioRobotics wins de novo nod from FDA for Aquabeam prostate treatment
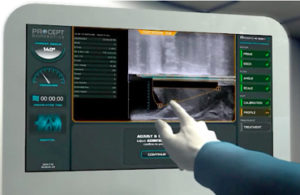
Procept BioRobotics said today that it won de novo clearance from the FDA for its Aquabeam device for treating benign prostate hyperplasia, saying it plans to have the device on the U.S. market early next year. Redwood Shores, Calif.-based Procept said the federal safety watchdog used the 181-patient Water study to inform its decision. The trial, comparing Aquablation…
Think Surgical wins CE Mark for TKA procedures with TSolution One robotic platform
Orthopedic robotic surgical platform developer Think Surgical said today it won expanded CE Mark approval in the European Union for its TSolution One surgical system, now cleared for use in total knee arthroplasty procedures. The TSolution One system is designed to aid in creating personalized pre-surgical joint replacement plans using CT-based 3D planning and open…
Feds launch 3-year innovation zone program for testing drones
The drone industry got a big boost from federal officials yesterday with a White House order to establish a three-year pilot program to test drones in partnership with state and local governments in specific jurisdictions. The Unmanned Aircraft Systems Integration Pilot Program was announced with President Trump directing U.S. Secretary of Transportation Elaine Chao to…
iRobot reports strong Q3, beats analyst estimates
Massachusetts-based iRobot (NSDQ:IRBT) today reported a 34% surge in third-quarter revenue and easily beat analysts’ consensus earnings projections. The company posted third-quarter profits of $22 million, or 76¢ per share, on revenues of $205.3 million for the three months ended Sept. 30, for a bottom-line gain of 13.21% on sales growth of 21.8% compared with Q3…
Renishaw wins FDA approval for neurosurgery planning software
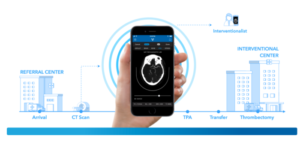
Renishaw, a British engineering company, received approval by federal officials to sell the latest version of its neurosurgery planning software in the U.S. The FDA granted 510(k) clearance for Renishaw’s neuroinspire software that blends MRI and CT datasets into a 3D volume, enabling neurosurgeons to identify and outline regions of interest. The approach is designed…
NIOSH to launch Center for Occupational Robotics Research
The National Institute for Occupational Safety and Health said this week it is launching the Center for Occupational Robotics Research. The newly founded center aims to explore the potential benefits and risks of robotic workers and develop appropriate guidance for safe interactions between humans and robots. “Robots working collaboratively with humans present a new workplace…
TransEnterix CEO Pope, riding high on FDA nod, eyes the laparoscopic surgery market
Share prices for TransEnterix (NYSE:TRXC) have more than doubled since the robot-assisted surgery company won an important clearance from the FDA last week and CEO Todd Pope is optimistic about his companies chances against the undisputed market leader, Intuitive Surgical (NSDQ:ISRG). Morrisville, N.C.-based TransEnterix, which acquired Senhance for $100 million in September 2015, finalized its clearance bid last…
TransEnterix wins FDA nod for Senhance robot-assisted surgery platform
TransEnterix (NYSE:TRXC) said last week that it won 510(k) clearance from the FDA for its Senhance robot-assisted surgery platform. TransEnterix, which acquired Senhance for $100 million in September 2015, finalized its clearance bid last month after applying in April. “The clearance of the Senhance system in the U.S. is a milestone in the progress of robotics and is…
FAA expands drone flight restrictions to include U.S. landmarks
The Federal Aviation Administration expanded its drone flight restrictions yesterday to include major U.S. landmarks at the request of the U.S. national security and law enforcement agencies. The new regulation prevents drone flights within 400 feet of 10 Department of the Interior sites: the Statue of Liberty, Boston National Historical Park, Independence National Historical Park…
Mazor X surgery platform from Mazor Robotics wins CE Mark
Mazor Robotics (NSDQ:MZOR) said today that it won CE Mark approval in the European Union for its Mazor X robot-assisted surgery platform, setting the stage for strategic backer Medtronic (NYSE:MDT) to put the device on the European market. “Receipt of the CE Mark is an important step in our plan to expand the patient, surgeon and hospital benefits…
Globus Medical jumps on FDA nod for Excelsius GPS robot-assisted surgery platform
Globus Medical (NYSE:GMED) said today that it won 510(k) clearance from the FDA for its long-awaited Excelsius robot-assisted surgery platform, which is designed to integrate intra-operative CT and fluoroscopic imaging technologies. Audubon, Pa.-based Globus said the clearance covers minimally invasive and open orthopedic and neurosurgical surgeries, including screw placement in spinal and orthopedic procedures. The system won CE…
7 medtech stories we missed this week: July 21, 2017
From Second Sight’s South Korean market entry to Sanuwave’s Indonesian distribution deal, here are seven medtech stories we missed this week but thought were still worth mentioning. 1. Second Sight enters South Korea market Second Sight announced in a July 5 press release that it has entered the market in South Korea with the implantation…
Intuitive Surgical wins FDA nod for inguinal hernia repairs with da Vinci Xi
Update: Removed information claiming benefits of robotic hernia surgeries at request of Intuitive Surgical, which said that the data came from an inaccurate source and was not valid. Intuitive Surgical (NSDQ:ISRG) said today it won FDA clearance for inguinal hernia repairs with its da Vinci Xi robotic surgical system. Inguinal hernias occur when tissue presses through…
Mazor Robotics reveals Israeli regulatory probe
Mazor Robotics (NSDQ:MZOR) yesterday revealed a probe by Israel’s version of the U.S. Securities & Exchange Commission, sending share prices down today in early trading. Caesarea, Israel-based Mazor said the Israeli Securities Authority searched its offices and questioned some members of management in May, “in connection with an investigation held by the ISA.” “Mazor has not been informed…
Intuitive Surgical wins FDA nod for daVinci X platform
Intuitive Surgical (NSDQ:ISRG) said today it won FDA 510(k) clearance for its new da Vinci X robotic surgical system, with a planned release of the platform later this year. The newly cleared da Vinci X system offers access to multiple robotic-assisted surgical technologies at a lower price than previous models, the Sunnyvale, Calif.-based company said. The…