TransEnterix has won expanded indications from the Food and Drug Administration for its Senhance robot-assisted surgical system. The system is now cleared for hernia and gallbladder procedures.
There are approximately 760,000 inguinal hernia and 1.2 million laparoscopic cholecystectomy procedures performed annually in the U.S. With this clearance, the system’s total addressable annual procedures in the U.S. has more than doubled to over three million.
“This indication expansion immediately doubles the addressable market for Senhance in the US and validates our regulatory strategy to successfully add to our indications for use,” said Todd M. Pope, president and chief executive officer of TransEnterix. “These expanded procedures are commonly performed at over 95% of hospitals in the United States. We believe this indication expansion will significantly increase the applicability of Senhance to more institutions, particularly those with a busy general surgery practice.”
The news sent TransEnterix shares up 28.8% to $4.03 apiece today in pre-market trading.
Related: TransEnterix opens the doors to a competitive US surgical robotics market
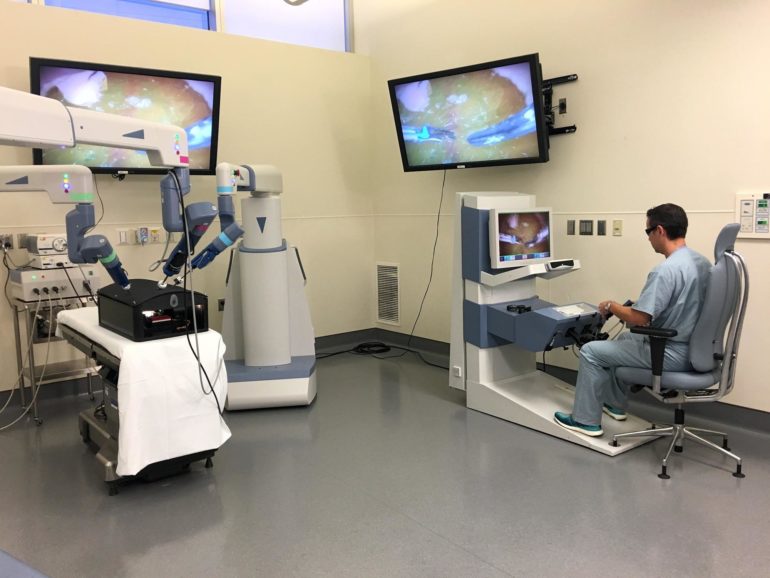
Florida Hospital in Orlando is the first U.S. hospital to use TransEnterix’s Senhance robot-assisted surgical system.
In the U.S., the system is now cleared for laparoscopic colorectal, gynecologic, inguinal hernia and cholecystectomy surgery. This enables Senhance to be used for some of the most common abdominal surgeries, including procedures in general surgery and gynecology.
“We have utilized Senhance broadly across a wide range of general surgery, upper GI surgery and colorectal procedures at our institution,” said Professor Dr. Frank Willeke, Chief of Surgery at St. Marien Hospital in Siegen, Germany. “We believe this procedural expansion for the US will allow surgeons there to incorporate the Senhance, as we have, as a highly-efficient, enabling and very promising technology that can impact the vast majority of surgeries commonly performed by general surgeons and their sub-specialties.”
Related: TransEnterix logs first U.S. sale
TransEnterix sold its first Senhance robot-assisted surgery platform in the U.S. in November 2017, exactly one month after winning FDA clearance. The sale, to Florida Hospital in Orlando, follows another sale in Taiwan made during the third quarter.




Tell Us What You Think!