|
Listen to this article
|
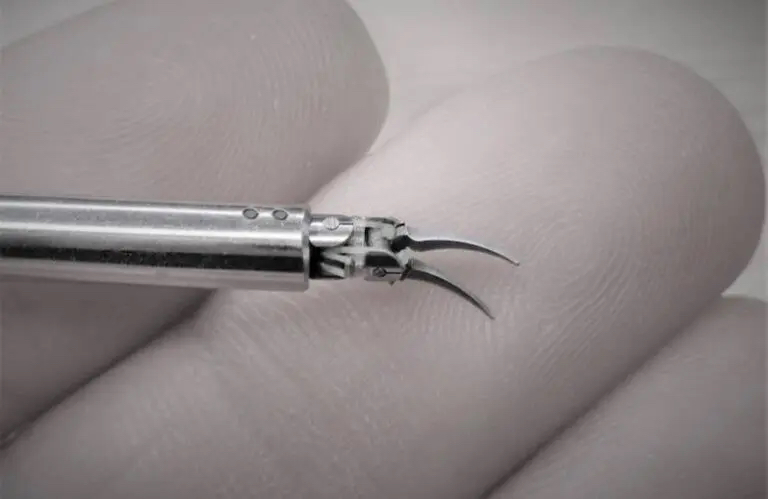
Image from Medical Microsintruments
Robotic microsurgery company Medical Microinstruments announced today that it raised $75 million in a Series B financing round.
Pisa, Italy–based Medical Microinstruments plans to use proceeds from the financing round, along with its planned U.S. presence, to move into its next stage of growth through expanded indications and ongoing commercialization efforts for its Symani microsurgery system.
The company designed Symani to address the challenges of microsurgery with the NanoWrist instruments for accessing and suturing small, delicate anatomy, such as veins, arteries, nerves and lymphatic vessels as small as 0.3mm in diameter. It provides motion scaling and tremor reduction to allow precise micro-movements.
Symani received CE mark in 2019, and the company intends to accelerate commercialization in the U.S. and Asia-Pacific, as well as advance clinical research through an FDA investigational device exemption (IDE) pivotal study.
“This financing round, coupled with our commitment to access the U.S. market and the addition of visionary leaders to our board, is an exciting moment for the surgical robotics space,” Medical Microinstruments CEO Mark Toland said in a news release. “We’re pleased to have bridged the Atlantic with premier U.S. life science investors, and existing European investors, who share our same vision of bringing microsurgical robotics to the world.”
Deerfield Management led the round, while the company received participation from new investors RA Capital Management and Biostar Capital, as well as existing investors Andera Partners, Fountain Healthcare Partners, Panakès Partners and Sambatech.
The company added Deerfield partner Dr. Andrew ElBardissi and RA Capital Management principal Tess Cameron to its board of directors. Additionally, former robotics business development manager for Comau, Arturo Baroncelli, will represent the company’s founders on the board.
Medical Microinstruments also announced a corporate redomicile from Italy to the U.S. Its recently opened Center of Excellence facility in Pisa, with 96 employees, will continue to be the hub of its research and development, manufacturing and other business activities.
“We are thrilled to add further depth and experience to the MMI Board and look forward to working with our new board members to build the robotic microsurgical space,” Andrew Cleeland, chair of the board, said.






Tell Us What You Think!