|
Listen to this article
|

AOT CEO Elmar Zurbriggen (right) and CFO Peter Oprandi with the company’s CARLO system | Source: AOT
Advanced Osteotomy Tools (AOT) announced that Elmar Zurbriggen will become CEO and Peter Oprandi will become CFO. Basel, Switzerland–based AOT developed a certified surgical device to cut bone with a laser. The system is called CARLO, for Cold Ablation Robot-guided Laser Osteotome.
“Elmar and Peter have both gained decades of experience as senior managers in the life science sector, working for global market leaders such as Johnson & Johnson, DePuy Synthes, AstraZeneca and Lonza,” AOT Chair Dr. Erich Platzer said in a news release. “We are delighted that they are now supporting AOT as CEO and CFO. Our goal is to improve patient care with innovative high-tech products. Elmar and Peter are exactly the right leaders for this.”
Zurbriggen was most recently an EVP at contract research, development and manufacturing firm Solvias and held VP roles at DePuy Synthes from 2008 to 2017, according to his LinkedIn profile. Johnson & Johnson acquired DePuy Synthes in 2012. It developed its own robot-assisted ortho surgery system, called Velys, after acquiring Orthotaxy and its orthopedic-surgery robot prototype in 2018.
Oprandi served as global head of finance, biologics, supply and operations at AstraZeneca from 2016 to 2019 and has held CFO and COO positions elsewhere since then, according to his LinkedIn profile.
“Our previous CEO, Cyrill Bätscher, decided to leave AOT in December,” Marketing Manager Sebastian Fischer said in an email today. “He was replaced by Christian Palme, who was functioning as managing director on an interim basis. Our former CFO, Martin Beckmann, decided to leave the company just recently. Today is his last workday at AOT.”
AOT was founded in 2010 by Dr. Alfredo E. Bruno, who served as CEO until 2018 and died unexpectedly in 2020. Bätscher took the role of CEO in 2018 after serving as COO.
AOT announced CE mark certification for CARLO in January 2021. The system is an alternative to piezoelectric bone cutters, oscillating saws and other mechanical instruments. A laser mounted to a robotic arm does the cutting, automatically guided along the cuts based on pre-operation navigation planning and simulations.
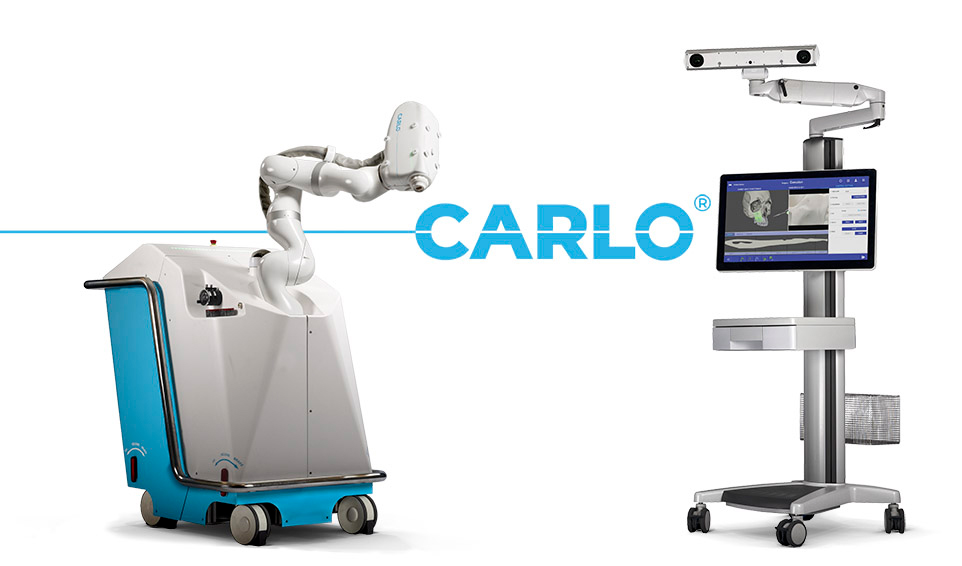
CARLO is Advanced Osteotomy Tools’ robotic laser bone-cutting system. | Source: AOT
“The laser pulse heats the water content of the bone in a fraction of a second, vaporizing it without heating the surrounding bone,” the company explains on its website. “The volumetric change and resulting kinetic energy fracture the bone structure on a microscopic level, and the excess energy ejects the debris. … Alternative trajectories can be defined or adjusted at any time during the procedure, and depth analyses serve to protect the underlying tissue by providing critical safety information to the surgeon.”
The company said the laser can cut custom shapes and interconnecting surfaces, cuts bone without causing carbonization, inflammation or necrosis, and allows for faster healing than mechanical instruments.
The device has not been cleared or approved by the FDA, and no ongoing clinical trials by the company were found in a search of the U.S. National Library of Medicine’s database of global trials.
“We submitted all necessary documents to the FDA early this year and are currently waiting for their approval,” Fischer said. “… Our ambition is to bring CARLO and therefore laser bone surgery to hospitals across the United States.”






Tell Us What You Think!