Neocis Inc. today said that it has received 510(k) clearance from the U.S. Food and Drug Administration for its Yomi system for robot-assisted dental implant surgery. The clearance allows for a new splint attachment, which broadens Yomi’s application to include full arch implant cases as well as partially edentulous cases, or those involving missing teeth.
The American College of Prosthodontists estimates that over 36 million Americans are missing all their teeth. The new Yomi edentulous splint will enable doctors to use Yomi’s technology to reach more of this population, said Miami-based Neocis.
“With this clearance, Yomi doctors will be able to leverage robotic technology to treat more patients and restore more smiles,” stated Alon Mozes, Ph.D. and CEO of Neocis. The company has received funding from Mithril Capital Management, Norwest Venture Partners, and robotic surgery industry pioneer Fred Moll.
Source: Neocis
Yomi the first dental robot to get FDA clearance
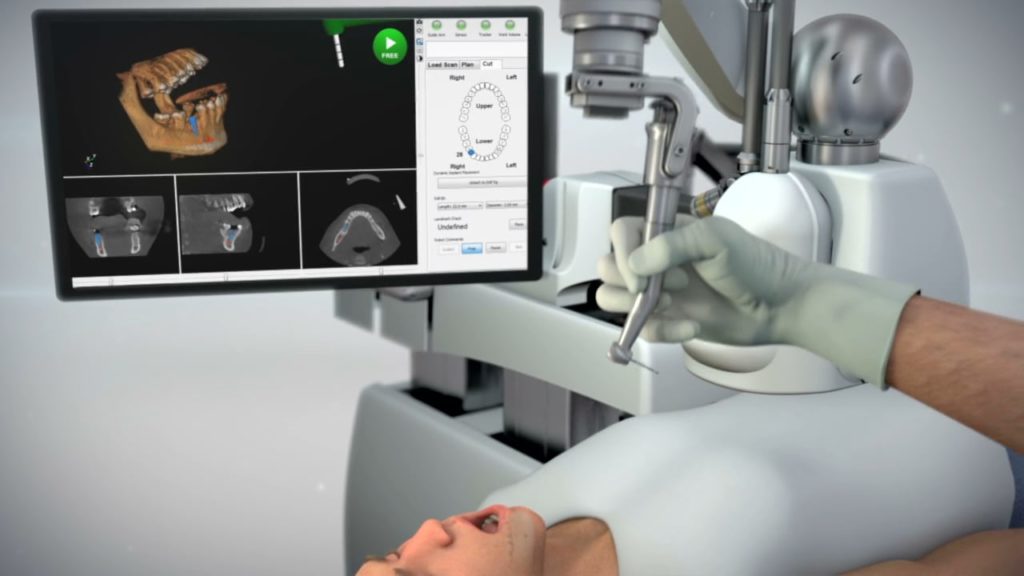
Neocis claimed that Yomi is the first and only FDA-cleared robot-assisted surgical platform for the dental industry. Yomi is a computerized navigational system intended to assist both the planning (pre-operative) and the surgical (intra-operative) phases of dental implantation.
The system enables surgeons to preoperatively plan implantation procedures and provides navigational guidance of the surgical instruments, said the company. Yomi is intended for use in partially edentulous and fully edentulous adult patients who qualify for dental implants.
Neocis said its system offers precise and accurate physical guidance through haptic robotic technology, which constrains the drill in position, orientation, and depth. The assistive technology provides the surgeon with complete control and, unlike plastic surgical guides, allows for clear visualization of the surgical site, it said.
Yomi can be used for flapless dental implants, which is a type of minimally invasive surgical approach. A flapless approach has been proven to lead to faster surgery, faster recovery, and less pain for the patient, said Neocis. The Yomi Robotics system has been used to place more than 2,000 dental implants across the U.S. In May, Yomi received FDA clearance for CT scanner compatibility.

Source: Neocis
Full arch testimonials
“The advent of Yomi’s full arch protocol is a dramatic shift in dental implant surgery,” stated Dr. Uday Reebye, M.D., DMD. A leader in oral and maxillofacial surgery with a practice in Durham, N.C., he was an investigator in the clinical study that supported the FDA clearance.
“A reduction in time to surgery, improved surgical efficiency and accuracy combined with a minimally invasive approach have made Yomi’s full arch protocol the gold standard,” he said. “Using Yomi, I was able to complete full arch surgery on average in 21 minutes.”
“I was so impressed by the accuracy of the first cases using Yomi robotic guidance in fully edentulous patients,” said Dr. Scotty Bolding, DDS, MS. The oral surgeon practices in both Fayetteville, Ark., and St. Petersburg/Tampa Bay, Fla., was also an investigator in the clinical study.
“The splints were extremely stable, easy to use, and remarkably accurate,” he said. “Additionally, there was excellent visibility in the surgical field that you cannot get with any available guide today. There is no doubt in my mind that Yomi will set the standard for the multi-implant cases in the future. I have been placing dental implants for over 30 years, and I have never experienced the precision or accuracy that was demonstrated with the new full arch splint.”




Tell Us What You Think!