 Procept BioRobotics said today that it launched a clinical trial aimed at winning reimbursement approval in France for its AquaBeam prostate treatment.
Procept BioRobotics said today that it launched a clinical trial aimed at winning reimbursement approval in France for its AquaBeam prostate treatment.
Redwood Shores, Calif.-based Procept said the 30-patient Francais Water study is designed to evaluate the safety and effectiveness of prostate ablation via waterjet in treating benign prostate hyperplasia in men aged 45 to 80. The primary outcome is total change in International Prostate Symptom score at 6 months, with a final data collection slated for August 2018, according to ClinicalTrials.gov.
“We successfully completed our first cases in September and are very encouraged by the procedural outcomes,” principal investigator Dr. Aurélien Descazeaud, of the Centre Hospitalier Régional Universitaire de Limoges, said in prepared remarks. “Aquablation is an exciting development for the treatment of BPH – an inconvenient, painful and often debilitating condition.”
Procept also said the first Aquablation cases were performed in Germany by Dr. Thorsten Bach of Asklepios Klinik Harburg in Hamburg.
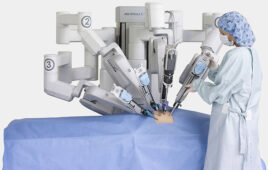
“Aquablation therapy is an innovative procedure that allows the surgeon to map the exact treatment contour on ultrasound, and then the robot autonomously resects the prostate tissue according to the surgeon’s plan, enabling it to be precise. With this device, our resection time is significantly reduced to less than five minutes, and patients will experience fewer sexual side effects than with traditional treatments,” Bach added. “We believe Aquablation has the potential to positively change the way we treat BPH.”
“Before Aquablation therapy, men had to choose between symptom relief and the high risk of sexual side effects. That is no longer the case,” president & CEO Nikolai Aljuri said. “It is our hope that this advanced treatment option will become more widely available throughout Europe, allowing men with BPH to experience the same benefits of surgical procedures like TURP, with less risk of side effects that impact their quality of life.”
Last month the company launched another study, Water II, an investigational device exemption study the company plans to use to back a bid for U.S. approval from the FDA.




Hi,
Aquabeam prostate therapy, my exhalted hope of human progress.Lets start together in Poland
Artur Pietrusa MD,Ph.D.FEBU
GeoMedical Hospital Katowice, Poland