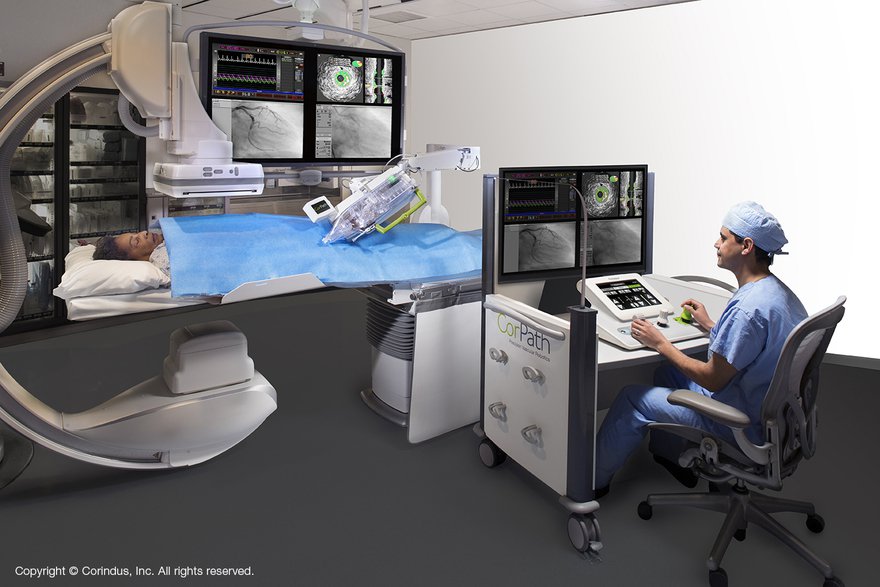
The CorPath GRX robotic surgical device from Corindus Vascular Robotics.
Corindus Vascular Robotics has won clearance from Japan’s Pharmaceutical and Medical Device Agency for its CorPath GRX robotic surgical device.
The CorPath GRX vascular robotics platform is designed to assist cardiologists in performing percutaneous coronary interventions (PCI) procedures, including angioplasty and stent placement. Japan is one of the largest markets in the world for PCIs, according to the announcement, with approximately 250,000 PCI procedures performed annually.
“I look forward to offering CorPath GRX to my patients during PCI, especially in complex procedures where precision and procedural control are extremely valuable. We are pleased to be one of the first Asia-Pacific countries to adopt this new and innovative technology,” Dr. Takafumi Ueno of Japan’s Kurume University Hospita said in a press release.
Last February, Corindus inked a Japanese distribution deal for its CorPath GRX device that included a $2 million advance and provision for 12 of the robot-assisted surgery platforms.
“There is strong demand for robotic-assisted intervention from key opinion leaders and the broad clinical community in Japan. PMDA approval of CorPath GRX in Japan represents a significant step in our ongoing effort to bring cutting-edge vascular robotics to global markets,” said Corindus president & CEO Mark Toland. “We believe robotics has the potential to enhance patient care in Japan by offering the benefits of robotic precision and advanced tools for physicians to perform these life-saving procedures.”
Corindus Vascular Robotic, based in Waltham, Mass., said it is pursuing reimbursement coverage for robotic percutaneous coronary intervention procedures in the region and has planned for a post-market study of the device.
Last month, Corindus saw shares fall after the robotic-surgery platform maker missed sales expectations on Wall Street with its first quarter 2018 earnings results. In March 2018, Corindus Vascular raised $25 million to support global commercialization of its CorPath GRX robotic surgical system.
It also that month received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the first automated robotic movement designed for its CorPath GRX platform. Called “Rotate on Retract” (RoR), the proprietary software feature is the first automated robotic movement in the technIQ Series for the CorPath GRX platform. It allows the operator to quickly navigate to a targeted lesion by automatically rotating the guidewire upon joystick retraction.




Tell Us What You Think!