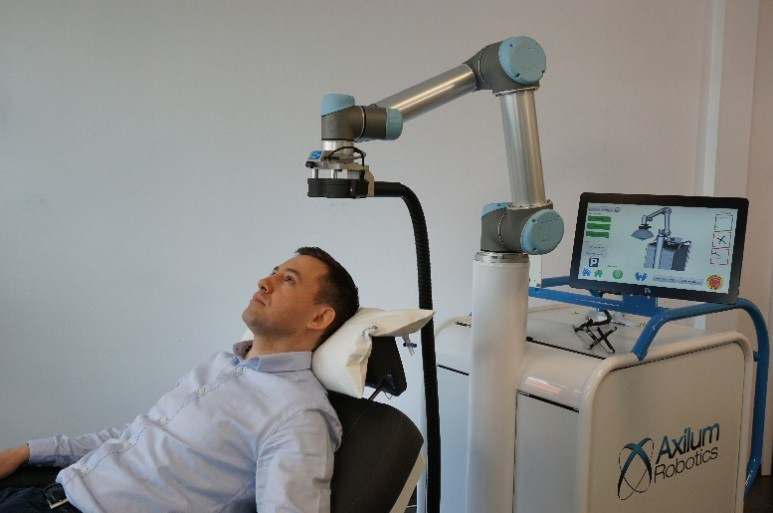
Axilum Robotics TMS-Cobot
Axilum Robotics, which specializes in the development of medical robots, announced that two weeks after the CE mark, it has received 510(k) clearance from the U.S. Food and Drug Administration to market the TMS-Cobot TS MV for the spatial positioning and orientation of the treatment coil of the MagVenture TMS Therapy system.
The company successfully developed and launched the TMS-Robot outside the U.S. It claimed that it was the first robot designed to assist health care professionals in delivering Transcranial Magnetic Stimulation (TMS).
Based on an invention of ICube laboratory in Strasbourg, Axilum Robotics has reinforced its expertise in medical robotics with the development of a new platform based on collaborative robot technology.
TMS-Cobot, the first medical device built on this platform, allows the company to offer an affordable and versatile system, thanks to a proprietary optical tracking system, which allows real-time control of the position, orientation, and contact of the stimulation device. It can also compensate for patient head motion.
The compatible MagVenture TMS Therapy System is indicated in the U.S. for the treatment of Major Depressive Disorder in adult patients who have failed to receive satisfactory improvement from prior antidepressant medication in the current episode. The FDA cleared the device for this use on July 31, 2015.
Axilum Robotics’ TMS-Cobot TS MV will be distributed in the U.S. by MagVenture Inc., which also distributes the MagVenture TMS Therapy System.
Transcranial Magnetic Stimulation is a rapidly expanding, non-invasive neurostimulation technique, particularly in the U.S., where TMS has been covered by health insurance since 2012. There is an increasing awareness of its current limitations when the coil is held manually or attached to a passive holder, requiring the patient to stay perfectly still.
“TMS-Cobot will allow us to better address the needs of the TMS therapeutic market by providing an affordable solution to allow precise TMS delivery, while relieving the operator from a demanding and time-consuming task and reducing the movement constraints on the patient,” said Michel Berg, CEO of Axilum Robotics. “It is important for the user to maximize the chance to deliver the stimulation dose at the right location and our medical robots help make this possible.”
“We are very happy to partner with MagVenture Inc.,” he added. “We are convinced that this robotized system represents a major step forward for the implementation of TMS in the U.S.A.”





Tell Us What You Think!