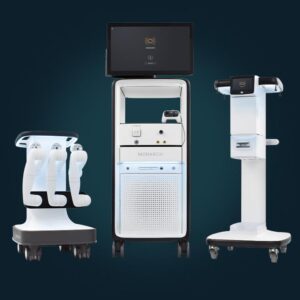
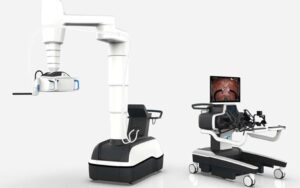
Johnson & Johnson MedTech said Monarch is its first robotic-assisted technology to receive regulatory approval in China.
Johnson & Johnson plans to submit Ottava surgical robot for FDA IDE next year
Johnson & Johnson first shared details on the Ottava surgical robotic platform nearly three years ago, and now its sharing its plans to seek FDA approval.
ReWalk Robotics solidifies Medicare coverage for personal exoskeletons
ReWalk Robotics announced that the Centers for Medicare & Medicaid Services (CMS) finalized a rule benefitting its exoskeleton technology.
First commercial cases completed with Levita Magnetics’ MARS surgical robot
Levita Magnetics announced the world’s first commercial use for its MARS magnetic-assisted robotic surgery platform.
CMR Surgical brings in $165M to support surgical robot
CMR Surgical announced today that it raised a funding round worth $165 million (£133 million) and hit a milestone with its surgical robot.
Virtual Incision raises $30M for mini surgical robot
Virtual Incision announced that it added a $30 million extension to its previous $46 million Series C financing round.
Moon Surgical picks up CE mark for updated Maestro surgical robot
The latest evolution of the Maestro System focuses on manufacturing scalability, together with increased capability and has simplified both usability and training.
Asensus Surgical targets FDA clearance for Luna surgical robot in 2025
Asensus Surgical announced its second-quarter financial results and shared an update on its Luna surgical robot platform.
Moon Surgical raises $55.4M to support surgical robot platform
Moon Surgical announced today that it raised $55.4 million and appointed a surgical robotics veteran as the chair of its board.
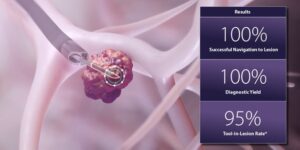
Noah Medical reports positive data for Galaxy surgical robot
The Galaxy surgical robot features advanced imaging technologies that provide real-time location updates for potentially cancerous lesions.
Alphatec acquires robotic navigation platform
Alphatec announced that it acquired all assets related to the REMI robotic navigation system from Fusion Robotics.
J&J’s Ethicon completes first robot-assisted kidney stone removal with Monarch platform
Auris Health, an Ethicon subsidiary, received FDA clearance for Monarch for endourological procedures in May 2022.
Ronovo Surgical unveils Carina surgical robot platform
Ronovo Surgical announced that it closed a new round of financing, bringing its total funds raised to more than $50 million since 2020.
Asensus Surgical wins CE mark for expanded machine learning
New features from Asensus Surgical include 3D measurement, digital tagging and image enhancement.
Intuitive expects da Vinci procedures to increase at least 12% in 2023
Intuitive Surgical released preliminary Q4 results that included a revenue miss, but the company expects da Vinci surgical robot procedures to grow.
Corindus rebrands to Siemens Healthineers Endovascular Robotics
Siemens Healthineers acquired Corindus for $1.1 billion in 2019. Corindus develops the CorPath robot-assisted device for coronary and peripheral vascular procedures.
Titan Medical to begin strategic review, may explore sale
Titan Medical announced today that it plans to review and evaluate strategic alternatives to unlock shareholder value.
Bionaut Labs raises $43M for drug delivery robots
Through magnetic propulsion, the company’s Bionauts can navigate the human body and deliver drugs locally.
Titan Medical seeks share consolidation to retain public listing
Surgical robotics company described share consolidation as the last option to regain compliance with Nasdaq’s $1 per share minimum bid price requirement and retain its listing.
Medical Microinstruments raises $75M for robotic microsurgery
Robotic microsurgery company Medical Microinstruments announced today that it raised $75 million in a Series B financing round.
Ekso Bionics exoskeleton cleared for use with MS patients
The latest generation of the Ekso Bionics platform, the EksoNR previously received clearance for stroke and spinal cord rehabilitation in 2016, then acquired brain injury (ABI) in 2020
FDA clearance expands indications for Xact Robotics’ ablation system
The Xact Ace system combines image-based procedure planning and real-time monitoring with hands-free precise robotic insertion and non-linear steering.